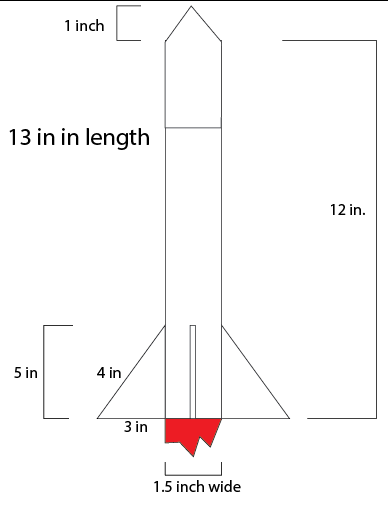
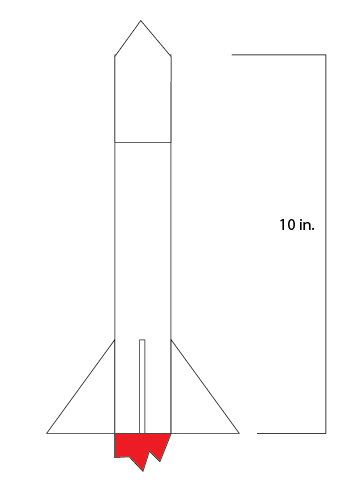
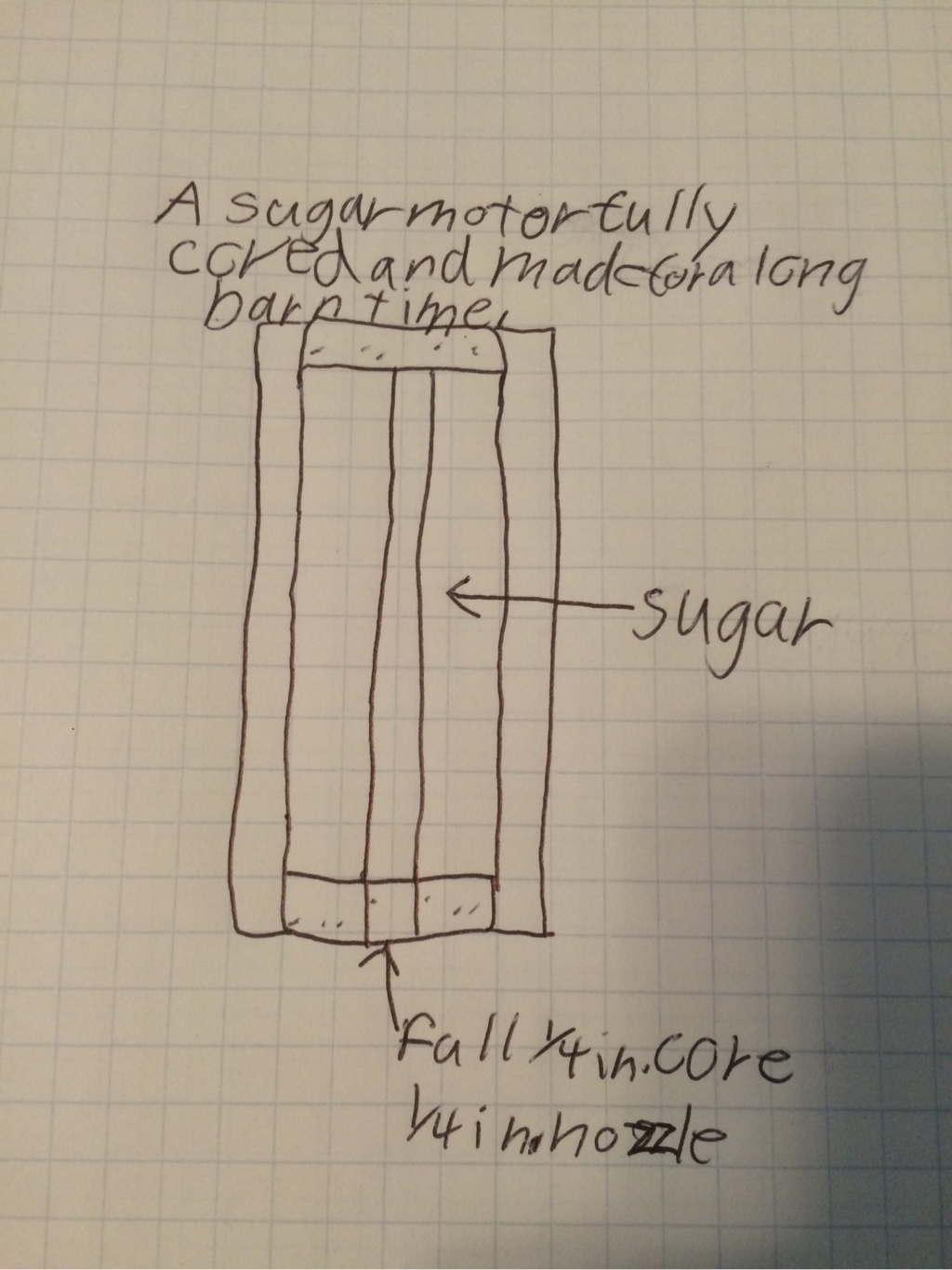
This is our rocket design and in this we wanted to keep things simple so we made the rocket length 12 inches like a ruler and a simple nose cone. We are having four regular triangle fins and are aiming for a lightweight rocket design. Garrett said we should have 4 fins to stabilize the rocket and move the center of mass. I contributed to making the body 12 inches and that is it.
|
-We are using the Kim Jong Un rocket motor
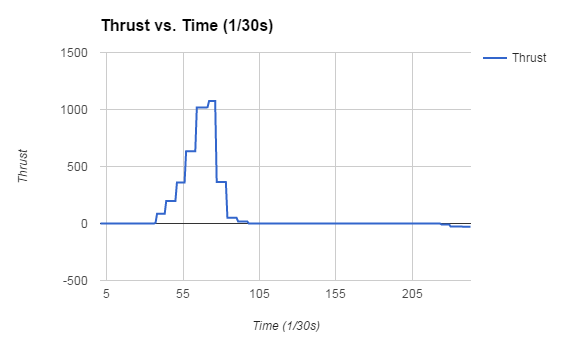
-We are making a relatively small rocket, only 10 inches in length not including the nose cone -I feel like this project is going to go well, we will see. Garrett and I are going to make this happen by using supplies from the classroom and getting started early. In this project I researched the two different kind of rockets, an end burner and a core rocket motor. The difference between the two is in theory cored creates more thrust because more fuel is being burned at one time. End burner's are very constant and reliable, they typically have a longer burn time than the cored rocket but sacrifice a lot of thrust which is fine for a smaller rocket. Then it was time to look at sugar rockets and black powder rockets. I chose a sugar rocket because I wanted to test something different and fully cored it. The black powder had a lot of benefits such as burn time but I wanted to try this out. Sugar rockets also seem like they don't have the chance to produce as much thrust as a black powder but Garrett and I wanted it to be for a smaller rocket. We finally gave Andrew our design and I was wondering whether our sugar rocket would blow up or not. I waited over the weekend and we heard bad news of tests gone bad conducted by Andrew which wasn't good considering he tested sugar rockets. Then the moment came when it was time to test the motor, we went out to the field and tested my motor and when I pressed the button to start it it screeched loudly. I thought it was going to explode but luckily it didn't and I felt a sense of relief. As you can see in the video it started off really strong and then just stayed alive for the rest of the time. We made a thrust curve in class for others to see how ours worked and I was thoroughly satisfied by our rocket's performance. A motor's thrust curve effects a rocket's performance because it shows how the rocket motor behaves and you can determine what kind of rocket it was. It had a total thrust of 14128g and a burn time of 7.4. Our motor was interesting because it got towards the end and didn't stop burning for some reason. It seems like a good design for a small or medium rocket because of its thrust time and overall thrust. In the end we liked Alex Wessel's groups rocket motor because Garrett and I wanted to make a smaller rocket and his didn't have too much thrust and an alright thrust time. The graph above is my sugar rocket and it surprised me with its performance and for this I am happy. Looking at this project I was very happy with the final product and enjoyed the project overall. I gained teamwork skills and got to understand rockets more in this project.
In this lab we were seeing how we could trap hydrogen inside a balloon by chemical reaction and then creating water. The way we started was by gathering all of our materials, some glass beakers and strips of metal. What happened in the experiment is we were introduced into how elements change if put under different temperatures and pressures and how an element can change from gas to liquid which is what we would be doing. I felt excited about this experiment because I wanted to see what was in store for the combustion part of it. Anyways we put magnesium I believe inside of water and put a balloon over the beaker right away. As you can see the balloon started to fill up with hydrogen, this was due to the water eating away from the magnesium taking out the hyrdogen and since hydrogen is a gas it filled the beaker and the balloon as well. It was an interesting reaction to see take place and the beaker's bottom felt really warm as well, the experiment so far was going really well and I was still curious as to what we would do afterwards. The chemical reaction I think that was taking place was somehow the hydrogen in the water and some element in the magnesium caused a surplus of hydrogen causing it to come out of the reaction. Garett and I were ready for the final part and Andrew pulled out a candle and I knew we were going to pop the balloon with it, so it was my turn to go up and held the balloon on the end of the stick above the small flame and I was very nervous and it popped in an amazing explosion and I was in awe. Shortly after Andrew showed us how water was formed inside of it and It was very cool. The experience was very cool, guided but made me feel like I was learning a lot so I didn't mind. I really enjoyed this lab and something I got from it was that blowing things up are fun and how elements can change depending on the temperature and pressure.
The earth science lab was recreating how the crust of the earth is made and how pretty much every rock inside the earth is made. We were replicating the natural course of how rocks inside the earth are formed on a basic level. We started off by learning about the 4 layers o the Earth which are the core, outer core, mantle, and crust. Basically everything in the earth's core and outer core is nickel and cobalt and the mantle is a mixture of those and silicate. The crust is the rocks formed by silicate and other elements. To start off the project Andrew brought out a couple of elements and pure water. He first measured the water and put some in a beaker and brought out a kind of cement mix or silicate and put the measured amount in the beaker as well. He then mixed the two together and I was somewhat confused but eager to see what would happen. After he mixed the two Garrett and I watched as Andrew but the mixture into separate vials, quite recklessly I might add, and gave them to each group. Then some groups and Andrew put the elements into the mixture and at first nothing was happening and then all of a sudden the red element shot up and created a little worm looking thing, and the white element created sort of jagged spikes on the bottom of the glass, it almost seemed unreal and that the elements were alive. What I thought was taking place was the element was bonding with the silicate and water and exchanging parts of each element and making the element solidify into some kind of formation. The sodium silicate bonded with the metal chloride, the sodium bonded with the chloride and the metal with the silicate causing a reaction. We watched for a little longer and I was thinking of how awesome our planet is and what takes place inside of it. This experiment was really cool and I want to see what the Crystals will look like next week but I'm not sure if Braden knocked them over today. I thought this experiment was really interesting because prior to this we hadn't learned about each science much in class so it was interesting to see this take place. Garret and I were pretty stoked to see this happening but the white element took some time to react than the other. What I gained from this is that elements inside the earth work perfectly together to make rocks, formations, and everything and that our earth is so amazing for it is what inhabits all human life and works perfectly on a large or small, however you want to look at it, scale.
|
Darian RoblesA mad chemist. Archives
December 2015
|